Key strategies to ensure your herd’s health
“No Herd Left Behind: Ensuring Immunity Onset and Duration for Optimal Health”
When it comes to protecting our loved ones and belongings, we often invest in insurance policies, analyzing the risks we might need to prevent or anticipate, and considering the duration of necessary protection. A clear example of this is travel insurance. When planning a trip, we know the start date and anticipate coverage for the entire duration.
The same goes for when we are planning Health Strategies for our herds. We consider the diseases (risks) that might affect our animals and take appropriate preventive measures against them. For this, it’s crucial to determine when we need the protection to start and how long it should last.
Vaccines are our insurance policy against diseases. To be as effective and protective as we need and expect, there are two critical attributes to consider: the Onset of Immunity (OOI) and Duration of Immunity (DOI).
The OOI determines how long the immune system can reach protective antibody levels after vaccination. With OOI, we know when our insurance policy starts, but how long does the protection last? To answer that question, we need to talk about the DOI, which is the moment in time at which vaccine-induced immunity begins to decline and provides less protection for a fully vaccinated animal.
Both OOI and DOI are specific to each vaccine since they are related to its antigenic power and ability to generate and maintain an immune response over a certain period.
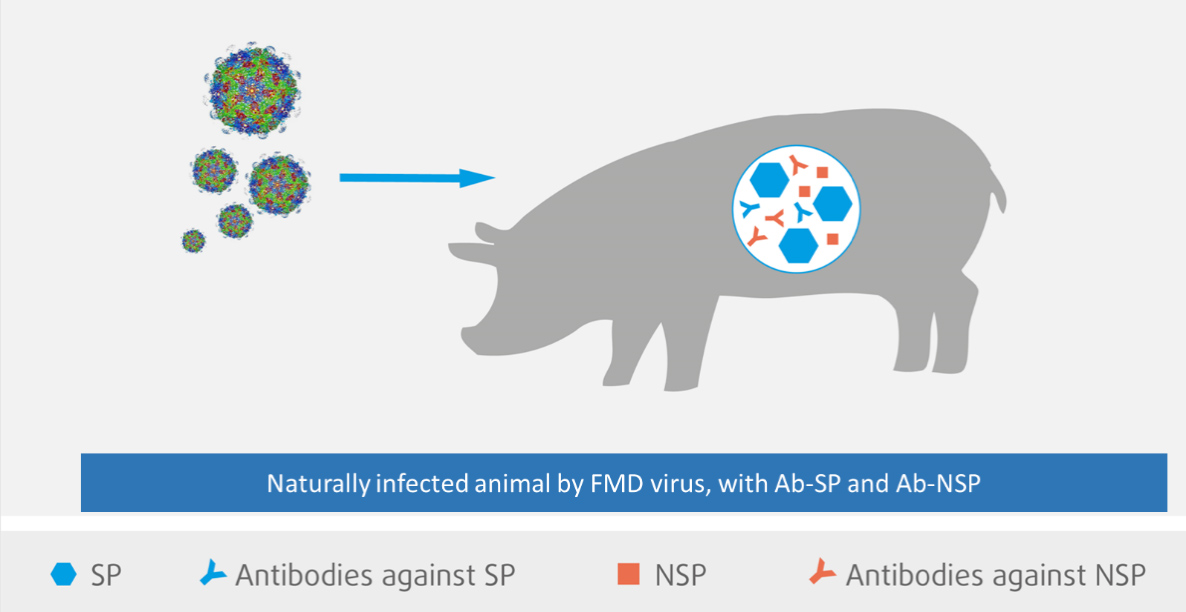
Immunity: The state of being immune, protected against an infectious disease conferred either by an immune response generated by immunization or previous infection.
This information is essential when designing an adequate Health Strategy for our herd. The induction of early protection is vital to prevent the spread of disease among susceptible animals, and the DOI helps us determine how long the herd will be protected, and whether a booster vaccination is needed.
Building a tailor-made health strategy for each farm, considering not only the risk assessment to prevent the main diseases that affect the herd but also the ability of vaccines to impact the immune system, is the best way to protect the animals and, in turn, their productivity. Furthermore, it allows the farmer to coordinate and organize his workload, and to schedule veterinary activities in advance.
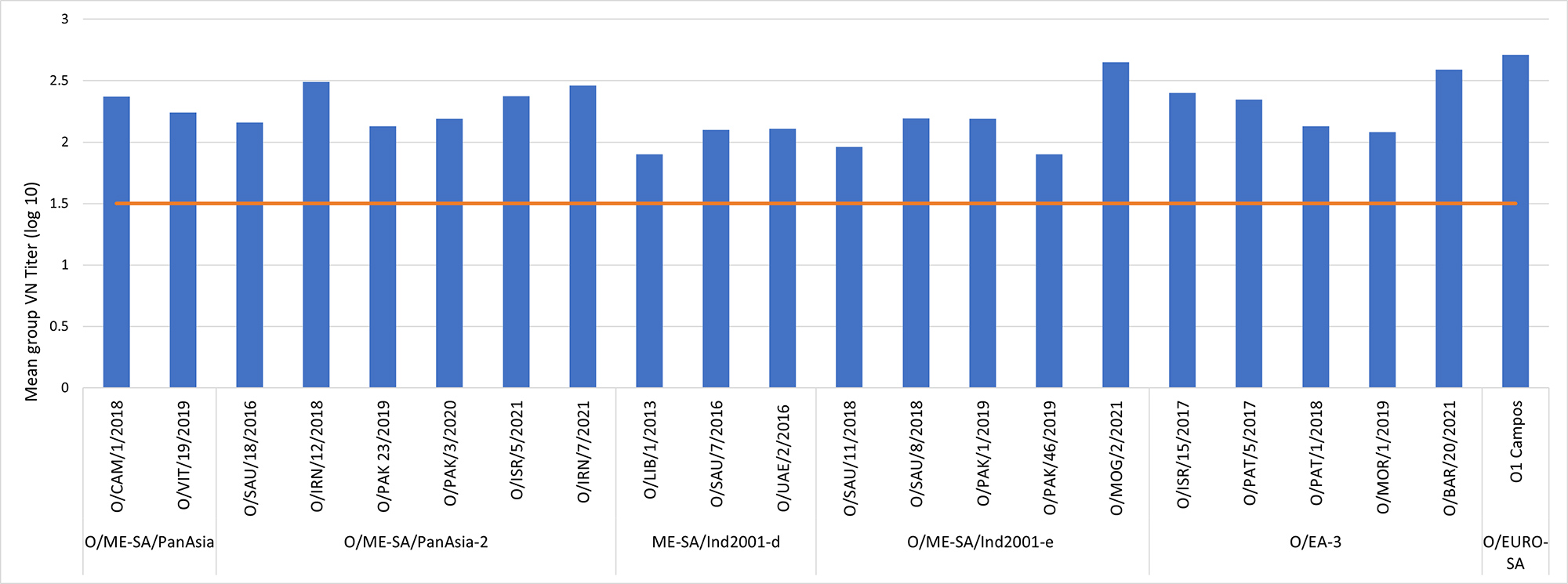
This information is available for Aftogen Oleo® and Bioaftogen®, where several clinical trials demonstrate an onset of immunity as early as 7 days and a duration of immunity of at least 180 days in primary vaccinated animals, even after a single shot.
In summary, when planning the most accurate Health Strategy for your herd, the most important pillars to consider are the risk assessment, to know what we should prevent, as well as the OOI and DOI of the vaccines chosen for this task. This helps avoid periods without protection, aiming to improve the herd’s health.
In your journey against FMD, World-Class vaccines are your best insurance policy.